As a life sciences marketing agency, we’ve seen a fundamental shift in how scientific buyers research and choose vendors. They no longer wait for sales calls. Now, they do the work themselves. They look up information online, compare different products, and check facts before they ever talk to a company.
This creates a problem. Most life science companies still use old ways to sell things. They show simple ads and send emails that have a low open rate. Then, they wonder why no one is buying or sales cycles take so long. There is a new way through life sciences content marketing to increase visibility on AI search like ChatGPT, Google, and other digital channels.
The answer is not more ads. It’s better content. Specifically, it’s life science lead generation services built on a content marketing foundation that earns trust, gets cited by AI, and shows up when buyers are actively researching solutions.
The Buyer Behavior Shift That Changes Everything
According to Gartner research, 61% of B2B buyers now prefer a rep-free buying experience. Even more telling: 73% actively avoid suppliers who send irrelevant outreach.
“73% of B2B buyers actively avoid suppliers who send irrelevant outreach.”
— Gartner, 2025 B2B Sales Survey
Source: Gartner Press Release
When looking at your pr and digital marketing for Life Sciences, it can come down to trust. If it’s a trust problem. Scientists, engineers, and procurement teams in life sciences have been burned by overpromising vendors. So they do their own research. They read application notes. They compare validation data. They check if your content shows up in AI answers.
That means your content is now your first sales rep. If it’s generic, compliance-risky, or hard to find, you lose before the conversation starts.

What Is Life Science Content Marketing?
Life science content marketing is the strategic creation and distribution of evidence-based content that builds trust, demonstrates expertise, and drives qualified pipeline for biotech, pharmaceutical, CDMO, and specialty chemical companies.
It differs from general B2B content marketing in three critical ways:
- Compliance requirements — Every claim must be truthful, balanced, and defensible under FDA OPDP guidelines and HIPAA marketing rules
- Evidence standards — Scientists expect data, not hype. Content must cite sources, show methodology, and acknowledge limitations
- Long sales cycles — Buying decisions take 6–18 months and involve multiple stakeholders, so content must serve researchers, engineers, procurement, and executives
According to the Content Marketing Institute, 74% of B2B marketers say content marketing generates demand or leads, and 49% say it directly generates sales or revenue. Yet only 22% rate their content marketing as extremely or very successful.
The gap is execution. Most teams lack the compliance workflows, AI optimization strategies, and distribution systems that turn content into pipeline.
See the RFP Agent Workflow (Life Sciences)
RFPs and security questionnaires steal weeks because answers live across sales, QA, regulatory, and legal. Our workflow drafts the first version from your approved technical documentation in minutes — then routes it for review when it matters. No made-up claims. No shortcuts. Just speed with guardrails.
Why This Changed in 2026: The AI Search Reality
Google’s search behavior has fundamentally shifted. According to Pew Research, 18% of Google searches now produce an AI-generated summary at the top of results. More importantly, users click a link in the AI summary only about 1% of the time.
Source: Pew Research Center, July 2025
This means your content must be cited, not just clicked. If AI tools like ChatGPT, Perplexity, and Google’s AI Overviews don’t quote your content, you’re not seen by the majority of modern buyers.
What makes content citeable?
- Short, clear answer blocks at the start of each section
- Structured lists and tables
- Credible sources and citations
- Consistent definitions
- High-quality visuals that support (not replace) text
We’ll cover the exact GEO (Generative Engine Optimization) services checklist later in this guide.

The Compliance Reality: What You Can and Can’t Say
Life science content marketing operates under stricter rules than most industries. The FDA’s Office of Prescription Drug Promotion (OPDP) sets the standard:
“Promotion must be truthful, balanced, and accurately communicated.”
— FDA OPDP Mission Statement
Source: FDA OPDP Overview
This applies to drug and device promotion. But even if you’re marketing lab services, reagents, or contract manufacturing, your buyers expect the same evidence standards.
HIPAA marketing rules also apply when protected health information (PHI) is used or disclosed for marketing purposes. According to HHS, marketing is defined as communication that “encourages recipients to purchase or use” a product or service, and authorization is generally required if PHI is involved.
Source: HHS HIPAA Marketing Guidance
How to stay compliant:
✅ Build an approved claims library with pre-vetted language
✅ Route all content through a review workflow (MLR, legal, compliance)
✅ Maintain an audit trail for every published asset
✅ Use “HIPAA-aware workflows” language, not “HIPAA-compliant” promises
✅ Never imply FDA endorsement
✅ Cite sources for every stat and claim
At Percepture, we maintain an in-house data compliance officer and use AI agents trained on approved materials to draft compliant first passes. This reduces review cycles from weeks to days.
Content Types Scientists Trust
Not all content formats work in life sciences. Scientists reward depth, precision, and transparency. Here are the 9 content types that consistently drive engagement and pipeline:
- Application Notes — Step-by-step protocols with real-world use cases
- Validation Stories — How a method was tested, what worked, what didn’t
- Method Comparisons — Side-by-side analysis of competing approaches
- Protocols and Checklists — Downloadable (or emailable) SOPs
- Teardown Guides — Deep dives into how a technology or process works
- Decision Matrices — Visual tools that map solutions to buyer needs
- Evidence Tables — Structured data with citations
- ROI and Risk Content — Executive-focused business case content
- Anonymized Case Studies — Real outcomes without revealing client names
Case Study: One integrated program we ran for a specialty life sciences client increased qualified leads by 382% in 120 days and increased qualified traffic by 67%. Another program for a CDMO increased qualified leads by 6x and LinkedIn leads by 4x in the first 120 days. Results vary by market and offer. These are examples from integrated programs.
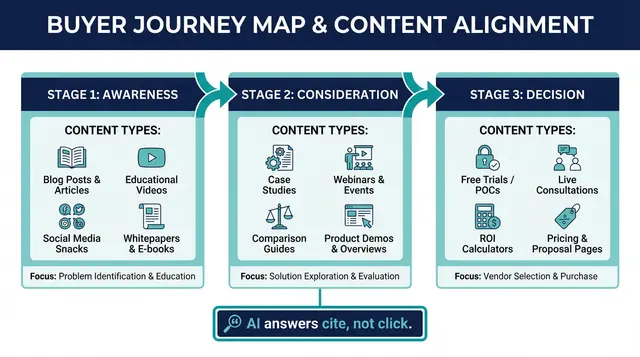
The Distribution Map for 2026
Creating great content is only half the battle. Distribution determines whether it gets seen, cited, and converted.
1. SEO + AI Overviews
Optimize for both traditional Google rankings and AI-generated summaries. Use structured data, internal linking, and answer-block formatting.
2. LinkedIn + Paid Social
Life science buyers are active on LinkedIn. Use organic posts, employee advocacy, and targeted ads to amplify reach.
3. Programmatic and Account-Based Targeting
Layer in display and programmatic ads to stay visible during long buying cycles. For more on this, see our guide on <a href=”https://percepture.com/life-sciences-insights/digital-marketing-for-life-sciences/”>digital marketing for life sciences</a>.
4. Digital PR Syndication
Earn backlinks and media mentions by syndicating content to industry publications, press release distribution, and earned media placements. This builds domain authority and increases AI citation probability.
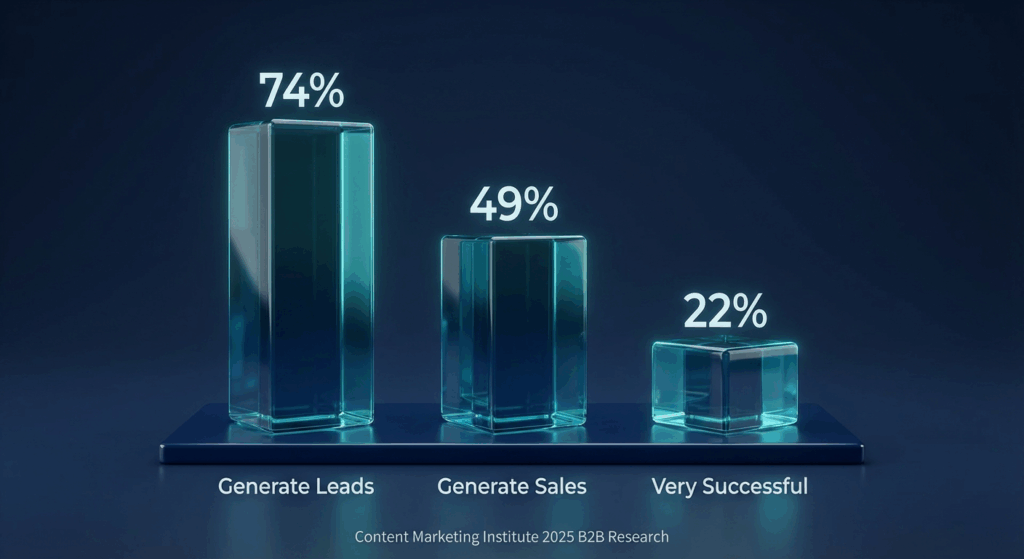
74% of B2B marketers generate leads from content, 49% generate sales, and only 22% rate their efforts as very successful
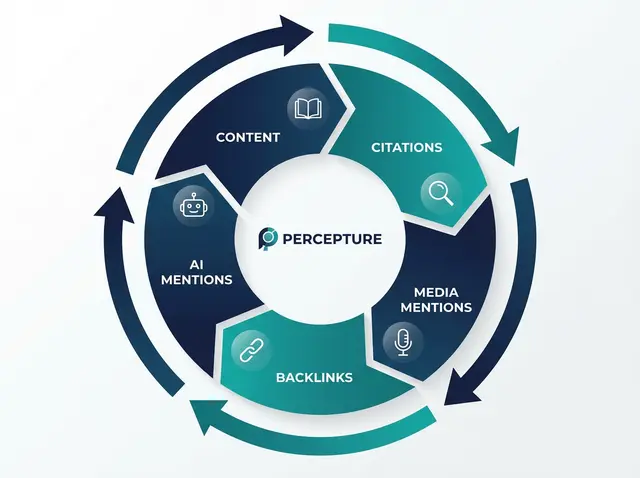
The Percepture Content Control Loop
Most agencies treat content as a one-time project. We treat it as a system. The Percepture Content Control Loop is a five-stage framework that connects evidence, compliance, distribution, authority, and pipeline.
Stage 1: Evidence
Start with data, not opinions. Use case studies, benchmarks, and vetted statistics.
Stage 2: Compliance
Route through your claims library and review workflow. Build an audit trail.
Stage 3: Distribution
Publish across SEO, AI search, LinkedIn, programmatic, and digital PR channels.
Stage 4: Authority
Earn backlinks, media mentions, and AI citations. Build trust signals.
Stage 5: Pipeline
Measure SQL rate, meeting volume, RFP velocity, and cycle time. Tie content to revenue. This loop is not linear. It’s iterative. Every piece of content feeds the next cycle.
Beyond GenAI: Agentic Workflows and the RFP Agent
Generative AI tools like ChatGPT are helpful. But they’re reactive. You ask, they answer. Agentic AI is different. It acts autonomously within guardrails you set.
At Percepture, we’ve built AI Sales Agents specifically for life sciences marketing and sales workflows. One example: the RFP Agent.
The RFP time problem:
According to Loopio’s 2024 RFP Trends & Benchmarks Report, the average team spends about 30 hours writing a single RFP response. QorusDocs research shows most teams take 6 to 10 days to complete an RFP, averaging 9.3 days.
Sources:
- Loopio RFP Trends Report
- QorusDocs RFP Survey
The RFP Agent workflow:
- Approved Materials — Agent pulls from your pre-vetted content library
- AI Draft — Generates a compliant first pass in minutes
- Review — Routes to your team for edits and approval
- Submit — Final version goes out faster, with full audit trail
Governance:
- No client data in demos
- HIPAA-aware workflows
- Audit trail for every output
This is not about replacing your team. It’s about giving them 20+ hours back per RFP so they can focus on strategy, not copy-paste.
For more on how we’ve applied AI to life sciences marketing since 2023, see our post on building the future of digital sales through AI marketing.

How to Get Cited in AI Search: The GEO Checklist
Google’s guidance on AI Overviews is clear: the same enterprise SEO services best practices apply. But there are specific tactics that increase your citation probability.
The 6-Point GEO Checklist:
✅ Allow crawling — Ensure robots.txt and meta tags don’t block AI indexing
✅ Make content findable via internal links — Build a strong internal linking structure
✅ Keep important content in text — Don’t hide key info in images or videos
✅ Support with high-quality visuals — Use charts, diagrams, and infographics
✅ Make structured data match visible text — Use JSON-LD schema that reflects actual content
✅ Use lists, tables, and short answer blocks — AI models prefer structured, scannable content

KPIs That Matter: Measuring Life Science Content Marketing
Most teams track vanity metrics like page views and social shares. Those don’t predict revenue. Here are the KPIs that do:
| Metric | Definition | Why It Matters |
|---|---|---|
| SQL Rate | % of leads that become sales-qualified | Shows content quality and targeting accuracy |
| Meeting Volume | # of discovery calls booked | Direct pipeline indicator |
| RFP Velocity | Time from first touch to RFP submission | Shorter cycles = better content |
| Cycle Time | Days from first touch to close | Content that educates shortens sales cycles |
| Pipeline Influence | % of closed deals that engaged with content | Proves content ROI |
According to McKinsey research, companies investing in AI for sales are seeing revenue uplift of 3% to 15% and sales ROI uplift of 10% to 20%.
The 90-Day Execution Plan
Here’s how to build a life science content marketing system in 90 days:
1 – Weeks 1–3: Audit + Foundation
- Audit existing content for compliance gaps
- Build your approved claims library
- Set up review workflow (MLR, legal, compliance)
- Identify top 10 keywords and search intent
2 – Weeks 4–6: Content Production
- Write 3–5 pillar articles (1,500+ words each)
- Create 2–3 decision matrices or comparison tables
- Develop 1 anonymized case study
- Optimize all content for SEO and AI search
3 – Weeks 7–9: Distribution + Authority Building
- Publish content on your blog
- Build internal links from existing pages
- Syndicate via digital PR (press releases, industry publications)
- Launch LinkedIn organic + paid campaigns
- Set up programmatic retargeting
4 – Weeks 10–12: Measurement + Optimization
- Track SQL rate, meeting volume, RFP velocity
- Monitor AI citations (ChatGPT, Perplexity, Google AI Overviews)
- A/B test CTAs and email capture forms
- Deploy RFP Agent for enterprise accounts
- Plan next quarter’s content calendar

Common Mistakes to Avoid
Even experienced teams make these errors:
❌ Gating everything — Scientists hate forms. Email content instead.
❌ Ignoring compliance — One bad claim can trigger an FDA warning letter.
❌ Writing for Google, not AI — If AI can’t quote you, you’re invisible.
❌ No internal linking — Orphan pages don’t rank.
❌ Vanity metrics — Page views don’t predict pipeline.
❌ One-and-done content — Great content needs ongoing promotion and updates.

Want a Content System Tied to Pipeline?
Book a 15-minute strategy audit. We’ll review your current content, rankings, and AI visibility — and show you exactly where the gaps are.
Frequently Asked Questions
1. What is life science content marketing?
Life science content marketing is the strategic creation and distribution of evidence-based content that builds trust, demonstrates expertise, and drives qualified pipeline for biotech, pharmaceutical, CDMO, and specialty chemical companies.
2. How is life science content marketing different from B2B content marketing?
It requires stricter compliance (FDA OPDP, HIPAA), higher evidence standards (data, citations, methodology), and longer sales cycles (6–18 months with multiple stakeholders).
3. Do I need FDA approval for my content?
Not for most marketing content. But all promotion must be “truthful, balanced, and accurately communicated” per FDA OPDP guidelines. Work with your MLR and compliance teams.
4. How do I optimize content for AI search?
Use short answer blocks, structured lists and tables, credible citations, consistent definitions, and high-quality visuals. Follow Google’s AI Overviews best practices.
5. What content types work best in life sciences?
Application notes, validation stories, method comparisons, protocols, teardown guides, decision matrices, evidence tables, ROI content, and anonymized case studies.
6. How long should a life science blog post be?
Aim for 1,500–3,000 words for pillar content. Shorter posts (800–1,200 words) work for specific how-tos or updates.
7. Should I gate my content?
No. Scientists hate forms. Use “email it to me” CTAs instead to capture contact info without friction.
8. How do I measure content marketing ROI?
Track SQL rate, meeting volume, RFP velocity, cycle time, and pipeline influence. Avoid vanity metrics like page views.
9. What is an RFP Agent?
An AI agent that drafts compliant RFP responses from approved materials in minutes, not hours. It maintains audit trails and routes drafts for human review.
10. How long does it take to see results?
Expect 60–90 days for SEO traction, 90–120 days for AI citations, and 120–180 days for measurable pipeline impact.
Connect with us today!
About the Author
Bob Generale Bob Generale is a partner of Percepture, where he leads digital PR, SEO, Paid Ads and AI search strategy for organizations operating in high-stakes, regulated markets—especially life sciences. His work centers on one objective: building measurable demand without compromising compliance, credibility, or brand integrity.
Since 2004, Bob has combined PR-led authority with search-led conversion strategy to help Fortune 500 teams compete for attention in crowded categories. He is a recognized voice in Generative Engine Optimization (GEO) and has published extensively on AI’s effect on telecom, data centers, and life sciences marketing since 2023, including practical frameworks for compliance-safe content, reputation management, and AI search visibility. He co-invented Search Incentive Marketing (SIM) and serves as an advisor to multiple technology and life sciences organizations, and is on the Board of the Ferrari Club of America, helping to lead the fastest-growing chapter in the USA.





